|
Other important groupings of elements in the periodic table are the main group elements, the transition metals, the lanthanides, and the actinides. Metals are lustrous, good conductors of electricity, and readily shaped (they are ductile and malleable), whereas solid nonmetals are generally brittle and poor electrical conductors. They are separated by a diagonal band of semimetals. Metals are located on the left of the periodic table, and nonmetals are located on the upper right. Semimetals exhibit properties intermediate between those of metals and nonmetals. The elements can be broadly divided into metals, nonmetals, and semimetals. Some of the groups have widely-used common names, including the alkali metals (Group 1) and the alkaline earth metals (Group 2) on the far left, and the halogens (Group 17) and the noble gases (Group 18) on the far right. Elements that exhibit similar chemistry appear in vertical columns called groups (numbered 1–18 from left to right) the seven horizontal rows are called periods. It arranges of the elements in order of increasing atomic number. The periodic table is used as a predictive tool. As expected, semimetals exhibit properties intermediate between metals and nonmetals. Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. The vast majority of the known elements are metals. 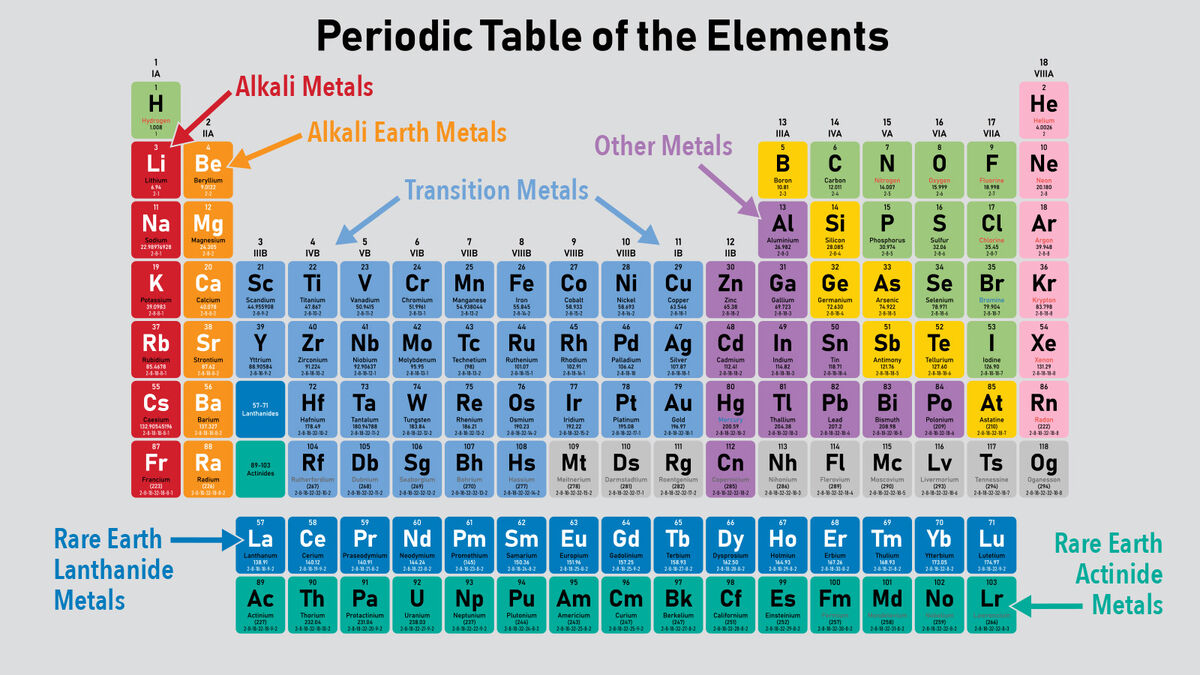
Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. The distinction between metals and nonmetals is one of the most fundamental in chemistry. Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. Gethin: So, the further down the table you go the more reactive they become.\) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line). So, the reason we didn't see the reactions of those last three metals is that they are so reactive the explosion would be far too big. Miss Armit: No, it's the second rarest element on Earth. That's not an element that you hear a lot of. We saw there lithium, then sodium and then potassium, but you didn't see rubidium, caesium and francium. As you can see the metal's get increasingly more reactive as you go down this group. Gethin: It's fair to say that you love the Group 1 metals, don't you. Well, sodium is below lithium in group one and the reactivity of the alkali metals increases as you go down the group, so potassium is even more reactive. So let's try it with another Group 1 metal - sodium.Īs you can see it reacts far more vigorously then lithium. 
And can you see the little purple trail there? That universal indicator is showing us that the water now contains an alkali in this case - lithium hydroxide. It's transferring its energy to its surroundings by heating. 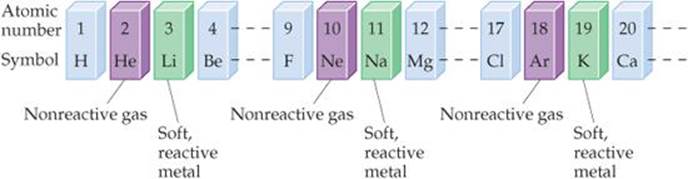
Well, you can see it's reacting with the water straight away - it's steadily fizzing away there. And if I put some universal indicator in, like this, you'll see that it's green and that means it's neutral.īut let's see what happens when we put the lithium in. And do you see how quickly it tarnishes, how quickly it's changing colour? Here we've got some water. 
Here I've got some lithium and I'm just going to cut a little bit off. When a Group 1 element reacts with water it produces a metal hydroxides solution and hydrogen gas. The elements with properties intermediate between those of metals and nonmetals are called metalloids (or. They're soft and shiny when freshly cut, but they quickly tarnish as the metal reacts with oxygen in the air. 2, metals occupy the left three-fourths of the periodic table, while nonmetals (except for hydrogen) are clustered in the upper right-hand corner of the periodic table. Let's take a look at some Group 1 metals then - also known as alkaline metals.įran: Group 1 elements are called the alkali metals. Gethin: And today we're going to be looking at the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
